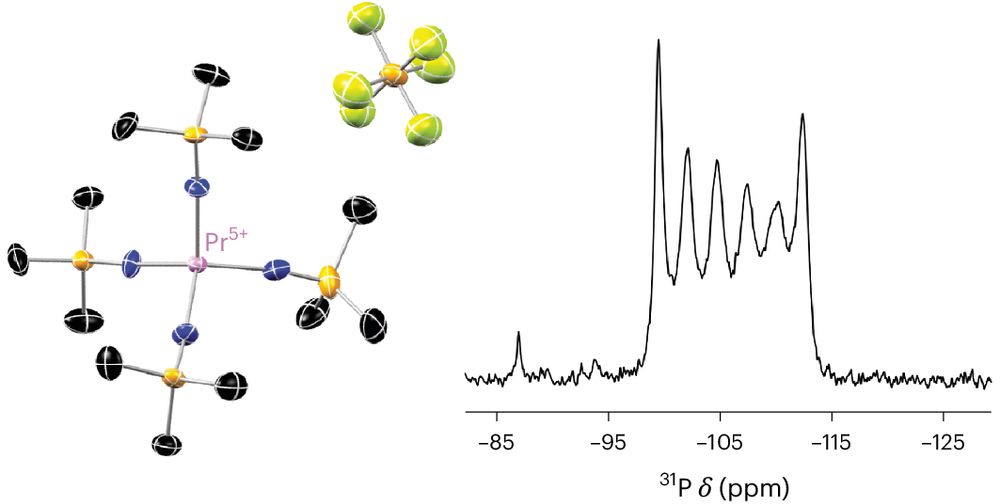
MOLECULAR ZINTL PHASES with tetra-anionic Si, Ge, Sn, Pb accessible using soluble form of Mg(0). Although all E(4-) anions are full valence-shell octets, they are highly reactive. Some are stable, some not. The Sn(-IV) complex is an 8e-reductant shorturl.at/InmD6 @natsynth.nature.com
03.03.2026 13:01
👍 24
🔁 4
💬 2
📌 0
Wow, this is beautiful…
03.03.2026 15:19
👍 1
🔁 0
💬 0
📌 0
Congrats, Fabian!
24.02.2026 23:13
👍 2
🔁 0
💬 1
📌 0

Thrilled to share the work of @clarencetankl.bsky.social and @antorna.bsky.social on elevating #sodium mediated deprotonative #borylation to #catalytic regimes with an insightful #mechanistic study in collaboration with Max Garcia Melchor @pubs.acs.org @unibe.ch
pubs.acs.org/doi/10.1021/...
29.10.2025 18:35
👍 46
🔁 11
💬 0
📌 2

ECAB member Sadig Aghazada @sadigaghazada.bsky.social
favorite @helvchimacta.bsky.social article: Willot et al. further develop a creative route to a broad family of multidentate and chiral NHC-Phosphine ligand precursors through methylenephosphonium adducts of manganese. doi.org/10.1002/hlca...
26.08.2025 13:05
👍 7
🔁 3
💬 0
📌 0
Continuing their tradition: another fantastic work from Liam @gliam96.bsky.social , Victor Mougel, and co-workers.
21.08.2025 16:40
👍 1
🔁 0
💬 1
📌 0
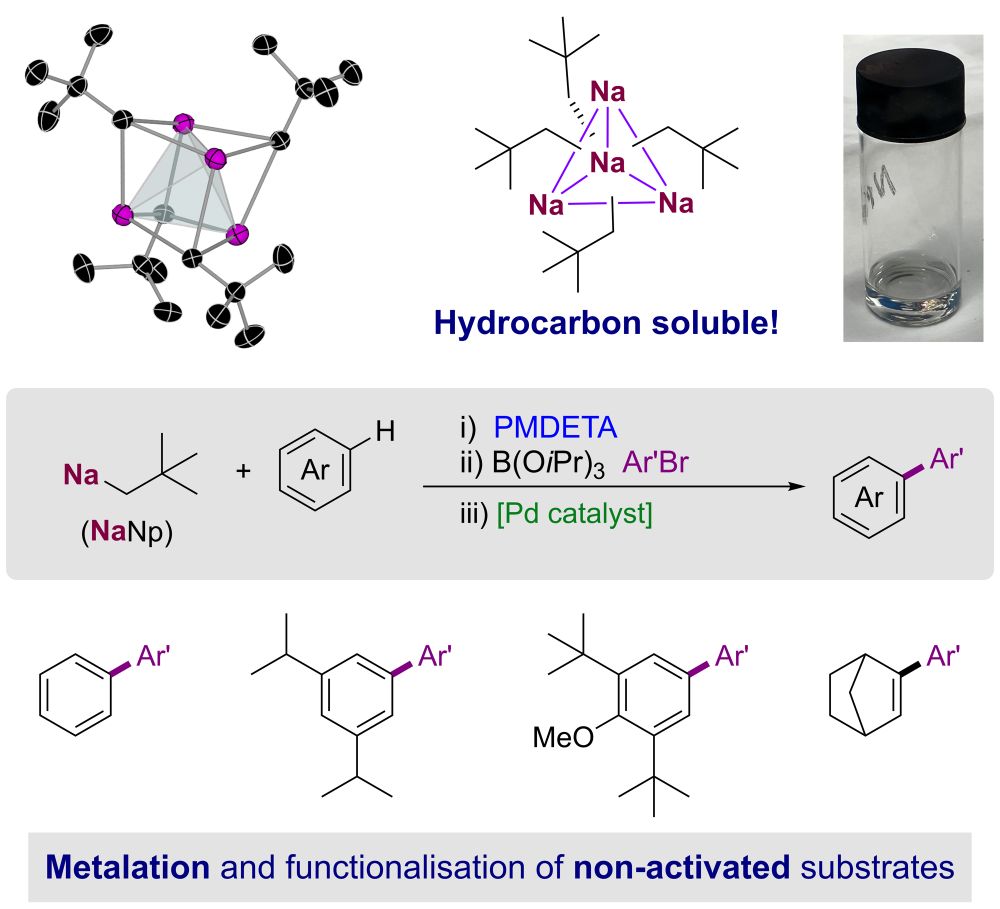
Excited to share the latest work from David Anderson just accepted @angewandtechemie.bsky.social on the structure and applications of #sodium #neopentyl a donor free #organosodium #reagent soluble in #hydrocarbon solvents @unibe.ch
onlinelibrary.wiley.com/doi/epdf/10....
29.07.2025 17:12
👍 56
🔁 11
💬 1
📌 1


Inspirational #KeynoteLecture by #KarstenMeyer @fau.de on the redox diversity of #ironChemistry including the isolation of an Fe (VII) complex!
07.07.2025 11:57
👍 29
🔁 6
💬 0
📌 1

Studienorganisation
Chiffre CHEM-PHARM-15179
We are hiring a new PhD student. If you love working with f-elements and early transition metals, plus living in a beautiful mountain area, please apply.
lfuonline.uibk.ac.at/public/karri...
23.06.2025 09:50
👍 9
🔁 5
💬 0
📌 0

Exploiting the similarity between Ca(II) and Yb(II), first Mg-lanthanide bonding has been realised. The peculiarities of this unique Mg-Yb bond are discussed. Mg(0)-Yb(II)? Mg(I)-Yb(I)? @JACS pubs.acs.org/doi/10.1021/...
04.04.2025 18:15
👍 32
🔁 5
💬 0
📌 0

We have open PhD positions @unibern.bsky.social
If you are interested in organometallic synthesis, transition metal and cluster chemistry, novel materials, and spectroscopy, this position is for you. Check the ad below and get in touch. #chemsky
Please re-quote and spread the news.
14.03.2025 17:50
👍 19
🔁 17
💬 3
📌 0
Thank you very much, Sven!
18.02.2025 08:16
👍 0
🔁 0
💬 0
📌 0

PREFORMED REDOX-ACTIVE INVERSE CROWNS: Finally out in @naturechemistry.bsky.social Highly selective reduction of N2O, epoxide, sulfur or O2. The ring can also be extended to a larger metalla-crown for stabilization of larger anions like N2O2(2-). Open access: shorturl.at/LZE6Y
17.02.2025 16:22
👍 58
🔁 11
💬 3
📌 0
Thanks a lot, Matle!
14.02.2025 14:04
👍 0
🔁 0
💬 0
📌 0
Thank you very much 😊
14.02.2025 14:04
👍 1
🔁 0
💬 0
📌 0
Thanks a lot, Xile!
14.02.2025 07:58
👍 0
🔁 0
💬 0
📌 0
Thanks a lot, Eva!
14.02.2025 06:43
👍 1
🔁 0
💬 0
📌 0
Yes, very exciting indeed, Dominik
13.02.2025 21:27
👍 1
🔁 0
💬 0
📌 0
Thanks a lot, Lisa)
13.02.2025 21:08
👍 0
🔁 0
💬 0
📌 0
Also, I am extremely grateful to Martin @albrecht-lab.bsky.social and Eva @evaheviagroup.bsky.social
for the very welcoming atmosphere in Bern. We will soon announce several PhD positions in beautiful Bern, so stay tuned.
13.02.2025 21:02
👍 5
🔁 5
💬 0
📌 0
I am thrilled to join the University of Bern
@unibern.bsky.social as a tt-asst. Prof. of Inorganic Chemistry this March. Very thankful to Karsten Meyer @fau.de, Christophe Copéret @coperetgroup.bsky.social,
@munzgroup.bsky.social, and all friends and family for their continuous support.
13.02.2025 21:02
👍 24
🔁 1
💬 7
📌 1

Transient Triplet Metallopnictinidenes M–Pn (M = PdII, PtII; Pn = P, As, Sb): Characterization and Dimerization
Nitrenes (R–N) have been subject to a large body of experimental and theoretical studies. The fundamental reactivity of this important class of transient intermediates has been attributed to their electronic structures, particularly the accessibility of triplet vs singlet states. In contrast, electronic structure trends along the heavier pnictinidene analogues (R–Pn; Pn = P–Bi) are much less systematically explored. We here report the synthesis of a series of metallodipnictenes, {M–Pn═Pn–M} (M = PdII, PtII; Pn = P, As, Sb, Bi) and the characterization of the transient metallopnictinidene intermediates, {M–Pn} for Pn = P, As, Sb. Structural, spectroscopic, and computational analysis revealed spin triplet ground states for the metallopnictinidenes with characteristic electronic structure trends along the series. In comparison to the nitrene, the heavier pnictinidenes exhibit lower-lying ground state SOMOs and singlet excited states, thus suggesting increased electrophilic reactivity. Furthermore, the splitting of the triplet magnetic microstates is beyond the phosphinidenes {M–P} dominated by heavy pnictogen atom induced spin–orbit coupling.
Our paper on triplet pnictinidenes (P, As, Sb) with transition metal substituents is out; congrats to Marc, Nils, Tarek and Rich and thanks to all that have contributed to get a comprehensive electronic structure picture @haenisch-group.bsky.social
pubs.acs.org/doi/10.1021/...
29.01.2025 15:50
👍 29
🔁 9
💬 4
📌 0