
Are you a clinician looking for a fully-funded postdoctoral research opportunity?
Check out our postdoctoral career development fellowships for clinicians, and see how you can join our vibrant community: www.crick.ac.uk/careers-and-...

Are you a clinician looking for a fully-funded postdoctoral research opportunity?
Check out our postdoctoral career development fellowships for clinicians, and see how you can join our vibrant community: www.crick.ac.uk/careers-and-...
Absolutely delighted that our latest paper is out today @natimmunol.nature.com. It has taken five years to uncover a role for cross-presentation in shaping the immune visibility of tumour antigens. A labour of love led by @drjonlim.bsky.social and Oliver Schulz. Great start of 2026!
I am deeply honoured to receive the Sue Eccles Early Career Investigator Award @metastasisresearch.bsky.social. Thank you for your continued support of emerging investigators in metastasis research!

🏆 MRS Sue Eccles Early Career Investigator Award: Jose M. Adrover

🚀 Two weeks to go!
The ECR Meeting brings together early-career scientists driving the next wave of metastasis research.
⚡ Don’t miss:
💡 Inspiring talks from rising leaders
💬 Community-led discussions
🌍 A global metastasis community
🗓️ Register by Nov 18 ➜ bit.ly/4qyxE0o
Program ➜ bit.ly/4qtpVR9

This review from Ambreen, @adrover.bsky.social et al. explores cancer-elicited changes to #neutrophil behaviour and proposes ways in which these may contribute to cardiovascular pathology. rupress.org/jem/article/...
📘 In #Cytokines collection: rupress.org/jem/collecti...
#Cytokines2025

My lab will be recruiting a PhD student this year. Have a look and apply if it sounds interesting!
adroverlab.com/open-call-fo...
We're now recruiting early career group leaders at the Crick to lead ambitious research programmes and explore bold scientific questions.
Hear our Director, Edith Heard, explain why the Crick is a unique place for curiosity-driven research.
Apply now ➡️ www.crick.ac.uk/careers-stud...

🔥Trio Fatale: Neutrophils, NETs and Necrosis
👉 authors.elsevier.com/a/1lk-k3qNrU...
We summarized for @cp-immunity.bsky.social
"Neutrophils drive vascular occlusion, tumour necrosis and metastasis" published in @nature.com by @megeblad.bsky.social @adrover.bsky.social rdcu.be/eFdau
Congrats!

A new model of #TumorNecrosis👹
Tumor CXCL1➡️myeloid skewing➡️
Ly6C-low Neutrophil forms intravascular #NeutrophilExtracellularTrap➡️
Intratumoral vascular occlusion➡️
Pleomorphic Necrosis-to-Metastasis
a role of TGFβ
@adrover.bsky.social @megeblad.bsky.social Nature 2025
www.nature.com/articles/s41...

Heart of the matter: #Neutrophils, #cancer, and #CardiovascularDisease. New review from Saira Ambreen, Afshan McCarthy, Andrés Hidalgo, and Jose Adrover @adrover.bsky.social @crick.ac.uk: rupress.org/jem/article/...
#TumorImmunology #CardiovascularBiology #InnateImmunity #inflammation

#Cancer patients display an elevated burden of #CardiovascularDisease. This review from Ambreen, @adrover.bsky.social et al. explores cancer-elicited changes to #neutrophil behaviour and proposes ways in which these may contribute to cardiovascular pathology. rupress.org/jem/article/...
Here's our take on how neutrophils can be a critical link between cancer and cardiovascular disease! "Heart of the matter: Neutrophils, cancer, and cardiovascular disease", just published @jem.org with Saira Ambreen, Afshan McCarthy and @drusqui.bsky.social
rupress.org/jem/article/...

Thanks to Cancer Discovery for highlighting our manuscript with such a nice summary!
Intravascular Neutrophil-Derived NETs Drive Tumor Necrosis and Metastasis aacrjournals.org/cdnews/news/...
@cdi-einstein.bsky.social Director @jaguirreghisolab.bsky.social co-leads a study showing how viral infections can trigger via IL6 signaling awakening of dormant breast cancer cells in lungs. CDI's research focuses in part on host variations controlling the timing of residual cancer awakening
Nature research paper: Neutrophils drive vascular occlusion, tumour necrosis and metastasis
go.nature.com/4lzilSb

Happy to share our latest preprint doing low cell number (mini-bulk) and single cell #proteomics on tumour associated neutrophils from human glioblastoma where we find multiple functional states that would be invisible to scRNAseq, some showing pro-tumoural states with potential therapeutic value
A fascinating study reports that neutrophils and NETs actively induce tumor necrosis. Congratulations, BDP @megeblad.bsky.social and @adrover.bsky.social on your latest publication in @nature.com.
As a new postdoc in my lab (@cshlnews.bsky.social → @jhu.edu) @adrover.bsky.social (now @crick.ac.uk) saw NETs in tumors—I shrugged. But he looked closer in 3D: NETs block tumor vessels, driving necrosis, hypoxia, EMT & metastasis.
🔗 www.nature.com/articles/s41...
@jhu-bdps.bsky.social @nature.com
This project started when I joined the @megeblad.bsky.social lab at @cshlnews.bsky.social, and continued at @hopkinsmedicine.bsky.social and at
@crick.ac.uk, with help from many colleagues along the way! It has been an absolute pleasure to work with Mikala and the rest of the team! (13/13)
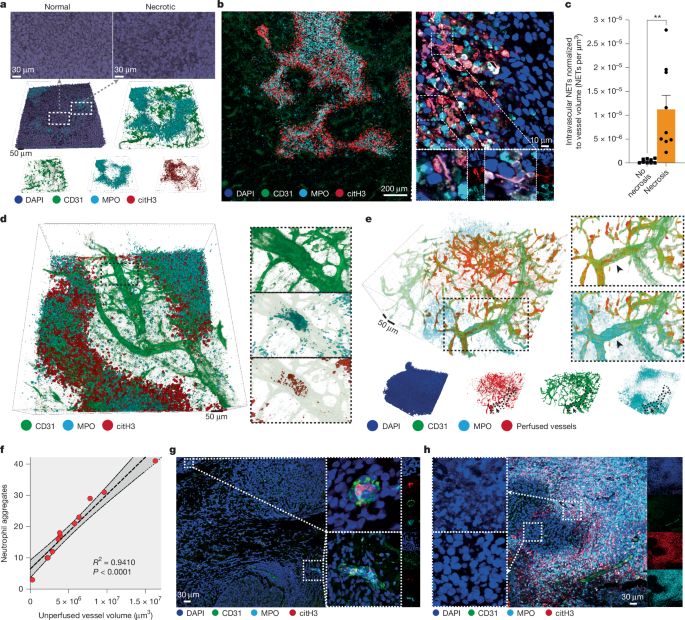
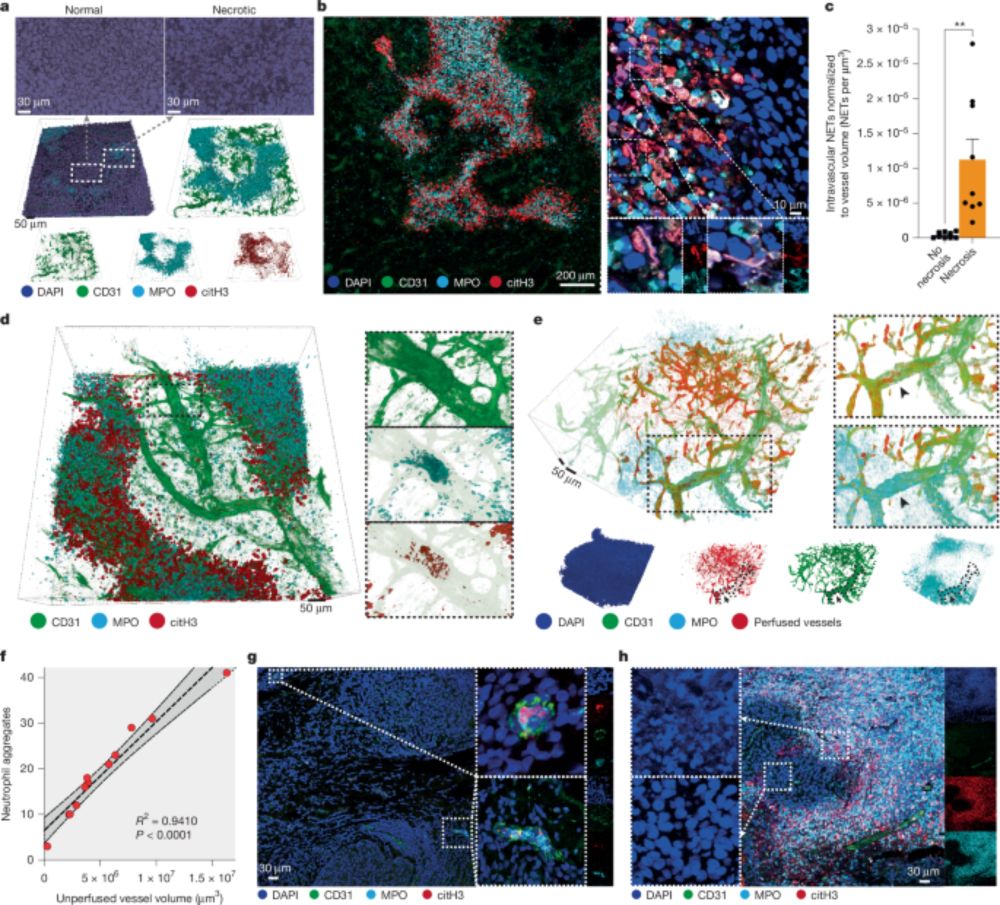
We show, therefore, that tumour necrosis is not a passive phenomenon, but an active phenomenon that we can target by targeting neutrophils or their NET formation ability. And by reducing tumour necrosis we can reduce metastatic spread. (12/13)

There, they form NETs and enter a feedforward loop with platelets that ultimately leads to the blockage of blood flow and subsequent necrosis of downstream tumour regions. This induces cancer cells around these necrotic regions to change and become more metastatic. (11/13)
In summary, we found that the tumour changes the haematopoietic compartment and drives the production of vascular-restricted neutrophils. Then, neutrophils leave the bone marrow and enter the bloodstream, eventually reaching the tumour vasculature. (10/13)

These neutrophils form NETs more efficiently than normal neutrophils, interact more with platelets, and, intriguingly, cannot extravasate and are stuck in the vasculature. We called them vascular-restricted neutrophils (vrPMNs). (9/13)
But why are neutrophils forming NETs in the vasculature? We found that the tumour communicates with the bone marrow and changes myelopoiesis to produce a novel neutrophil subpopulation. (8/13)

Using spatial transcriptomics, we found that tumour necrosis increased metastasis because cancer cells in peri-necrotic areas gained pro-metastatic traits in a TGFb-driven manner. (7/13)

We found that the process was driven by NETs. When we block NETs genetically or pharmacologically, tumour necrosis is greatly reduced. This, interestingly, also reduced metastatic spread. (6/13)
These aggregates were blocking blood flow in the tumour, and that caused the necrosis of downstream tumour regions. (5/13)

Interestingly, around necrotic regions we found vessels that contained intraluminal aggregates of neutrophils, NETs and platelets. (4/13)

We found that the architecture of necrosis was peculiar and intricate in different tumour models (we call it “pleomorphic necrosis”), and these regions were covered by neutrophils and NETs. (3/13)