Last days to apply this position in our group! Do join us in this great Institute @crg.eu, with great colleagues, and help shaping the science and culture of our lab!
Last days to apply this position in our group! Do join us in this great Institute @crg.eu, with great colleagues, and help shaping the science and culture of our lab!
Thank you A. Perez-Ramos and P. Bovolenta for this great perspective on our article about Optic Cup morphogenesis @dev-journal.bsky.social !
Congrats ☺️☺️
Fantastic interaction with @carldmodes.bsky.social
and @liormoneta.bsky.social , who did all the modelling. It was great to work in such an interchangeable setting between biology and physics, to lear about #ShapeProgramming and shed a little more light on how organs gain their 3D shape.
This work also had the great contributions of A. Szalapak,
@louise-dagher.bsky.social and
@the-chaotician.bsky.social . Special thanks also to
@lcferme.bsky.social for the stardist model! And of course to Caren and all the @nordenlab.bsky.social
Finally, we disrupted apical patterned behaviors experimentally. The optic cup completely failed to start the invagination! (side note, the phenotype was so striking that I thought the microscope had crashed while imaging)

Our model became even cooler: we converted tissue segmentations into networks. Like this we could test the strain patterns in the REAL geometry of the tissue

But can the shape transitions at the apical surface be sufficient to drive basal surface invagination? Updating our model with basal surfaces: apical strain patterns alone are enough to initiate invagination—even if basal surfaces are passive

First, we needed to understand how are the apical surfaces curving. We went back and forth between experiments and modelling (inspired by shape programming). We found each apical surface displays different cell behaviors, generating its own in-plane strain patterns,
But the surprise came from the apical surfaces. They undergo shape transitions before invagination starts. Could they be driving the process?
Separating the basal surface of the two epithelia (NE and RPE), we saw that they change their shape at the same time!
We followed 3D shape changes of the forming optic cup, by tracking the basal surface
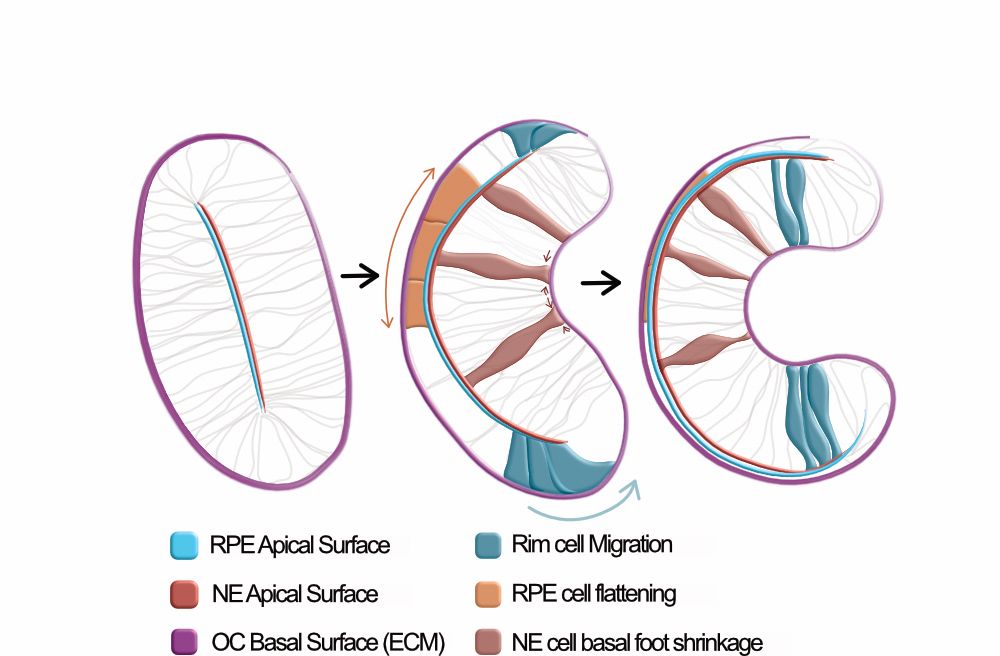
Many things happen at the same time: 2 epithelial layers differentiate, cells migrate, other cells flatten and others constrict their basal surface, and the lens grows in. We went through (a LOT of) negative results to try to understand which processes were triggering the onset of the invagination
We used the Optic Cup formation as a system. The shape transition from a flat vesicle to an hemispheric eye precursor is just mesmerizing
We departed from a bigger question: how 3D shape emerges in complex tissues, from the interaction of multiple cell behaviors and mechanical inputs?
Briefly, we found that OC invagination onset relies on active, collective cell behaviors, that create patterned strains at the apical surfaces. In addition, OC morphogenesis involves the cooperative reinforcement of two independent active patterns. More details bellow 👇

I am thrilled to share the main result of our work at @nordenlab.bsky.social with @liormoneta.bsky.social and @carldmodes.bsky.social . We bridged DevBiology + theoretical physics to understand how the eye becomes round #morphogenesis #devbio #biophysics #zebrafish www.biorxiv.org/content/10.1...